Abbott is initiating a proactive, voluntary recall of powder formulas, including Similac, Alimentum and EleCare manufactured in Sturgis, Michigan, one of the company’s manufacturing facilities.
The recall does not include any metabolic deficiency nutrition formulas.
Consumers are asked not to use Similac, Alimentum, or EleCare powdered infant formulas if:
- the first two digits of the code are 22 through 37; and
- the code on the container contains K8, SH or Z2; and
- the expiration date is 4-1-2022 (APR 2022) or later.
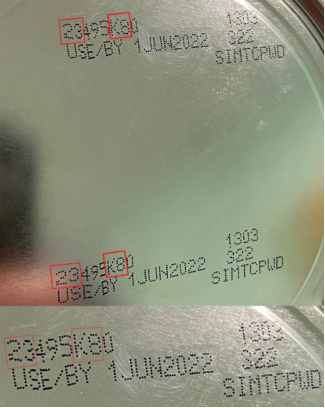
Consumers are urged to visit similacrecall.com to find out if their product is affected.
Further information can be found on the following link: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/abbott-voluntarily-recalls-powder-formulas-manufactured-one-plant
GDBS
























